Recently, the Minister of Health of Vietnam promulgated Circular 13/2021/TT-BYT on the issuance of circulation numbers and import of medical equipment for COVID-19 prevention and control in urgent cases, especially, stipulating 17 medical equipment to fight against COVID-19 quickly issued with circulation numbers

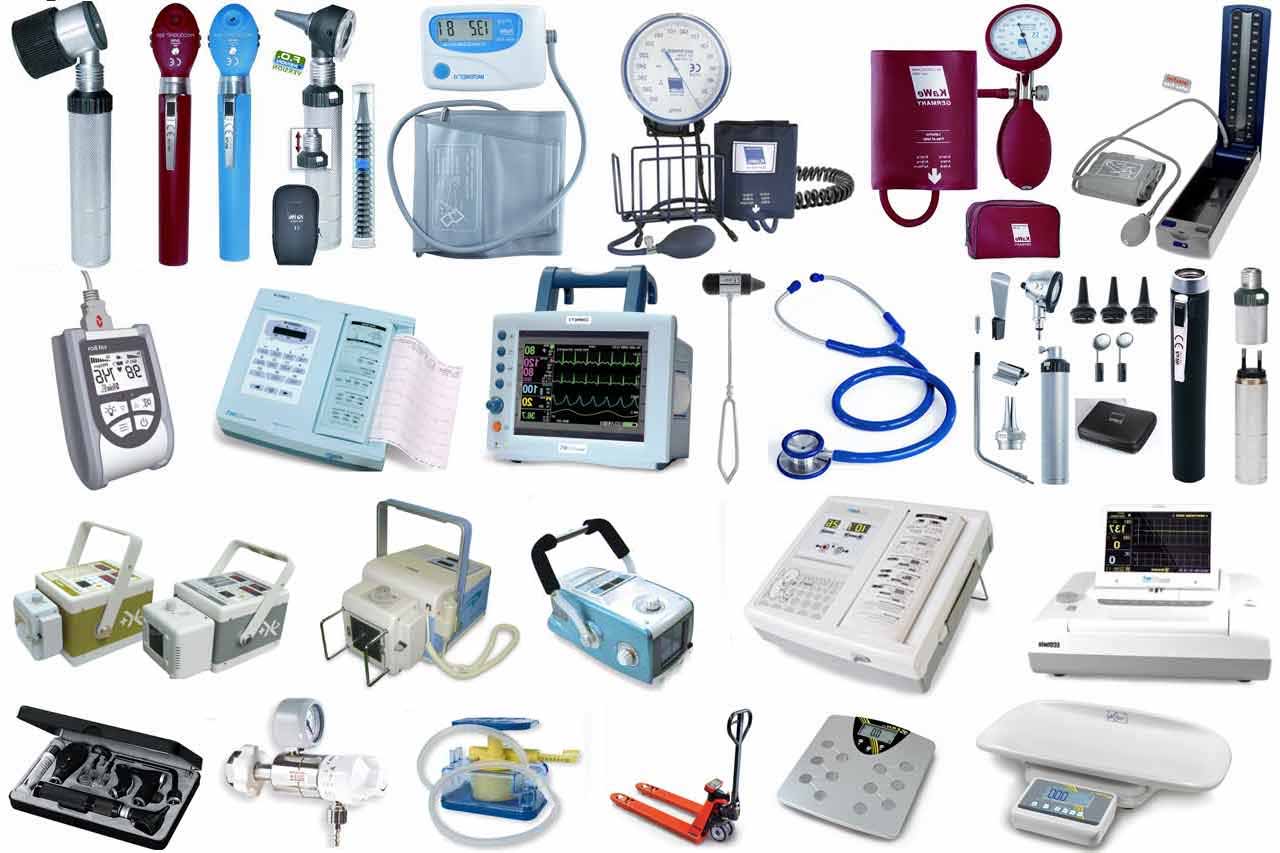
17 medical equipment to fight against COVID-19 quickly issued with circulation numbers in Vietnam
Specifically include:
- Extraction machine;
- PCR machine;
- Chemicals (biological products) running the SARS-CoV-2 PCR test machine;
- Rapid test kit for antigen/antibody against SARS-CoV-2;
- High-function ventilators, invasive and non-invasive ventilators, non-invasive ventilators, high-flow oxygen machines, portable ventilators;
- Continuous dialysis machine;
- Portable X-ray machine;
- Color Doppler ultrasound machine ≥ 3 probes;
- Blood gas meter (measuring electrolytes, lactate, hematocrite);
- Patient monitor ≥ 5 parameters;
- Electric injection pump;
- Infusion machine;
- Pacemaker defibrillator;
- ECG machine ≥ 6 channels;
- Portable ultrasound machine;
- Coagulation time meter;
- Hemodynamic meter.
The Ministry of Health of Vietnam stipulates that medical equipment may apply the form of fast issuance of free-sale registration numbers if it simultaneously meets the following conditions:
(1) Belonging to the list specified in Appendix 1 issued together with Circular 13/2021/TT-BYT ;
(2) Belongs to one of the following cases:
- Approving for circulation or emergency use by one of the following organizations: US Food and Drug Administration (FDA) - USA; Therapeutic Goods Administration (TGA) - Australia; Health Canada (Health Canada); Japan's Ministry of Health, Labor and Welfare (MHLW) or Pharmaceuticals and Medical Devices Agency (PMDA) - Japan;
- Be approved by the competent authorities of the European countries specified in Appendix 2 to Circular 13/2021 for circulation and emergency use;
- Belongs to the list of emergency-use SARS-CoV-2 testing products announced by WHO on its website at https://extranet.who.int (Coronavirus disease (COVID-19) Pandemic —Emergency Use Listing Procedure (EUL) open for IVDs | WHO - Prequalification of Medical Products (IVDs, Medicines, Vaccines and Immunization Devices, Vector Control););
- Belonging to the list of popular products for testing for SARS-CoV-2 issued by the European Health Security Committee (EUHSC) published on its website at https://ec. europa.eu (Technical working group on COVID-19 diagnostic tests | Public Health (europa.eu)
- Be granted a commercial import license in Vietnam before September 16, 2021;
- Be produced domestically in the form of technology transfer, for medical equipment falling into one of the above cases;
- Be produced domestically in the form of processing for medical equipment in one of the cases mentioned above.
Note:
- The circulation number of the issued medical equipment includes:
+ The certification number of the Department of Medical Equipment and Medical Works on the standard announcement document, made according to form No. 1 specified in Appendix 3 issued together with Circular 13/2021 of Vietnam, for medical equipment economy of categories B
+ Number of circulation registration certificate for medical equipment issued by the Department of Medical Equipment and Medical Works according to the form specified in Appendix 4 issued with Circular 13/2021 for medical equipment economy of categories C, D.
- Value of circulation number: The issued circulation number is valid in Vietnam from September 16, 2021 to December 31, 2022.
Bao Ngoc
 Article table of contents
Article table of contents





.Medium.png)
.Medium.png)
.Medium.png)
.Medium.png)
